KAEA1111 Assignment – Material Balance Calculations
Deadline: 20th April 2026, 17:00.
Submission Method
Scan and upload your handwritten answers to PDF format in Spectrum.
General Instructions
There are ten (10) questions. Answer ALL questions.
- Show all steps of calculation clearly – marks will be awarded for method, not just the final answer.
- Typewritten and handwritten answers only. Ensure they are neat, easy to read, and organized with proper units and final answers.
- Use proper unit conversion, significant figures, and material balance principles.
- Upload answers in PDF files. Write name, and matrix number on top left of answer file.
Question 1 (8 marks)
The effective heat capacity of a gaseous mixture is given by
𝐶𝐶𝑃𝑃 = 7.13 + 0.577 × 10−3𝑇𝑇 + 0.0248 × 10−6𝑇𝑇2
Where 𝐶𝐶𝑃𝑃 is in 𝐵𝐵𝐵𝐵𝐵𝐵 ∙ 𝑙𝑙𝑙𝑙 𝑚𝑚𝑚𝑚𝑙𝑙−1 ∙ ℉−1 and 𝑇𝑇 is in ℉.
a. What are the units for the coefficients (7.13, 577 × 10−3, and 0.0248 × 10−6, respectively)?
b. Change the equation in the form in which 𝐶𝐶𝑃𝑃 is given in
𝑘𝑘𝑘𝑘𝑘𝑘𝑚𝑚𝑚𝑚𝑙𝑙. 𝐾𝐾 and 𝑇𝑇 is in 𝐾𝐾.
Question 2 (7 marks)
The heat capacity of ammonia over a limited temperature range is given by the expression below
𝐶𝐶𝑃𝑃 = 0.487 + 2.39 × 10−4𝑇𝑇
𝐵𝐵𝐵𝐵𝐵𝐵
Where 𝐶𝐶𝑃𝑃 is in 𝑙𝑙𝑙𝑙𝑚𝑚℉ and 𝑇𝑇 is in ℉.
a. What are the units for the coefficients (0.487 and 39 × 10−4, respectively)?
𝐽𝐽
b. Determine the expression for 𝐶𝐶𝑃𝑃 in 𝑔𝑔℃ in terms of 𝑇𝑇 in ℃.
Question 3 (10 marks)
Methane and oxygen react in the presence of a catalyst, to form formaldehyde. In a parallel side reaction, some of the methane is oxidized to carbon dioxide and water.
𝐶𝐶𝐶𝐶4 + 𝑂𝑂2 → 𝐶𝐶𝐶𝐶𝐶𝐶𝑂𝑂 + 𝐶𝐶2𝑂𝑂
𝐶𝐶𝐶𝐶4 + 2𝑂𝑂2 → 𝐶𝐶𝑂𝑂2 + 2𝐶𝐶2𝑂𝑂
The feed to the reactor contains equimolar amounts of methane and oxygen. Assume a basis of 100 mol feed per second.
a. Draw and label a flow chart.
b. Use material balances to derive expressions for the product stream component flow rates in terms of the two extent of reaction.
c. The fractional conversion of methane is 0.900 and the fractional yield for formaldehyde is 0.855. Calculate the molar composition of the reactor output stream and the selectivity of formaldehyde production relative to carbon dioxide production.
Question 4 (20 marks)
A liquid mixture containing equal mass fraction of ethanol (MW=46.07) and water (MW=18) is fed into a distillation column. The product emerged at the top stream contain 95 wt. % of ethanol, while bottom product flow rate is measured at 600 lbm/h. Given the inlet flow rate of the liquid mixture is 1500 lbm/h.
(a) Sketch a completely labelled process flow diagram for the above process.
(4 marks)
(b) Analyze the degree of freedom of this process.
(3 marks)
(c) Determine the mass fraction, mole fraction and molar flow rates for both ethanol and water in the bottom stream.
(8 marks)
(d) Calculate the amount of fresh feed (lbm/h) if the production rate of ethanol at overhead stream is targeted to achieve 25 metric ton/day with the operation hour of 18 h/day.
(5 marks)
Question 5 (20 marks)
Two aqueous sulfuric acid solutions containing 20.0 wt% H2SO4 (SG = 1.139) and 60.0 wt% H2SO4 (SG = 1.498) are mixed to form a 4.00 molar solution (SG = 1.213).
(a) Calculate the mass fraction of sulfuric acid in the product solution.
(b) Taking 100 kg of the 20% feed solution as a basis, draw and label a flowchart of this process, labelling both masses and volumes and do the degree-of-freedom analysis. Calculate the feed ratio (litres 20% solution/litre 60% solution).
(c)What feed rate of the 60% solution (L/h) would be required to produce 1250 kg/h of the product?
Question 6 (10 marks)
Powdered milk is produced using spray drying process where the liquid milk is injected into a chamber through a spray nozzle, resulting the liquid milk becoming mist. Hot air is injected from the bottom of the dryer. The milky mist is then contacted with hot air, where most or all the water in the mist evaporates and the dried milk solids fall to a conveyor belt at the bottom of the chamber. A simplified block diagram to produce powdered milk is shown in Figure 1.
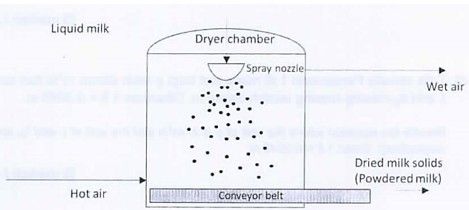
Figure 1
In the factory of “AnDad Essential” milk company, powdered milk is produced in a spray dryer which has a diameter of 6 m and height of 6 m. The liquid milk fed to the spray nozzle contains 70% water by mass, the rest is milk solids. Hot air enters the dryer at 167 °C and 730 mm Hg absolute. All moisture is evaporated during contact with hot air. The outlet gas, i.e. the wet air flows at 311 m3/min containing 12 mol% water and leaves the dryer at 83°C and 1 atm absolute. Assume ideal gas behaviour for the gas flows.
The given information below might be useful:
Molar mass of water: 18 g/mol.
Molar mass of air: 29 g/mol.
Gas constant, R = 0.08206 m3·atm / (kmol·K); and 1 atm = 760 mm Hg.
(a) Completely label a block diagram based on the given information.
(b) Determine the mass of liquid milk fed to the dryer.
(c) Determine the production rate of powdered milk.
(d) Determine the volumetric flow rate of inlet hot air.
Question 7 (15 marks)
A stream of complex agricultural raw materials is fed into a bioreactor that will be operated continuously. The compositions for the elements involved (C, H, N, O, P and S) in the 50 ton/h feed stream are known. However, the exact biochemical reactions inside the bioreactor are uncertain. There are two reactor effluent streams, one solid and one liquid. All N and P are trapped in the solid stream. By stating a 1000 ton as basis, perform a degrees-of-freedom analysis for the bioreactor.
Question 8 (10 marks)
Paper pulp is sold on the basis that it contains 12% moisture; if the moisture exceeds this value, the purchaser can deduct any charges for the excess moisture and deduct for the freight costs of the excess moisture. A shipment of pulp became wet and was received with a moisture content of 22%. If the original price for the pulp was $40/ton of air-dry pulp and if the freight is $1.00/100 lb shipped, what price should be paid per ton of pulp delivered?
Are You Searching Answer of this Question?
Request Malaysian writers to write a plagiarism-free copy tailored to your question.
Get Help By Expert
If you are struggling to complete your KAEA1111 Chemical Engineering Process Analysis I Assignment (University of Malaya), especially with material balance calculations, unit conversions, or process flow diagrams, you can choose Assignment Helper like many other students. Our experts provide chemical engineering assignment help tailored to your university requirements with accurate and well-structured solutions. You can also review our University of Malaya assignment example to check the quality of our work. Order our online homework helper service today and receive a 100% custom, human-written, and plagiarism-free solution.
Need the complete answer?
